Test Lal

Dr Lal Pathlabs One Of India S Largest Blood Test Labs Exposed Patient Data Techcrunch

Comparative Evaluation Of Pyrogens Tests In Pharmaceutical Products

Pyrostar Es F Series Wako Lal System

Lal Reagents For Lal Assays Charles River

Coronavirus Symptoms Testing Centre Near Me Faq Coronavirus Dr Lal Pathlabs

Histopathology Test Report Dr Lal Pathlabs Blog
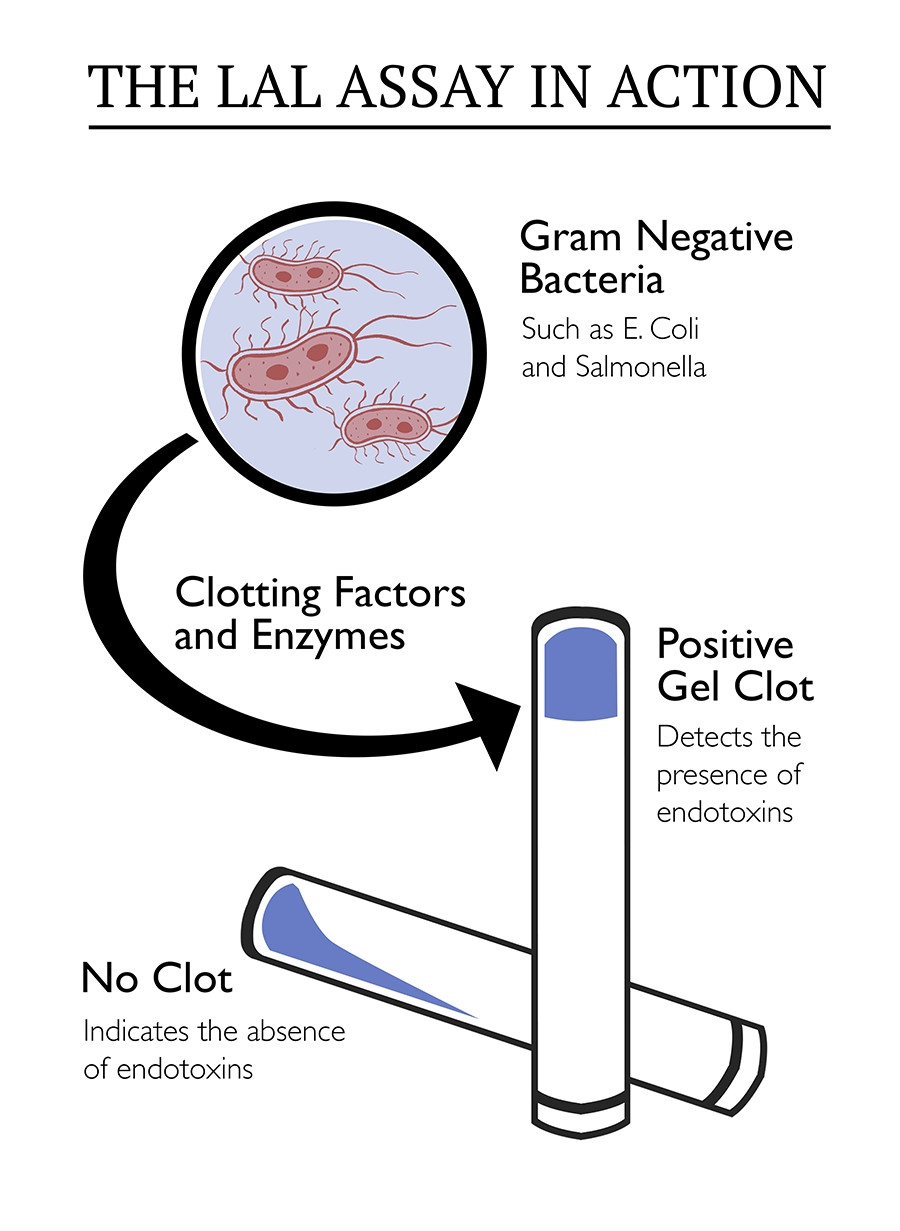
LAL Reagents Introduction to the LAL Test Limulus Amebocyte Lysate (LAL) tests detect and quantify bacterial endotoxins extracted from the outer membrane of gram negative bacteria The critical component of the LAL reagents used in endotoxin tests is derived from blood cells (amebocytes) of the horseshoe crab, Limulus polyphemus.

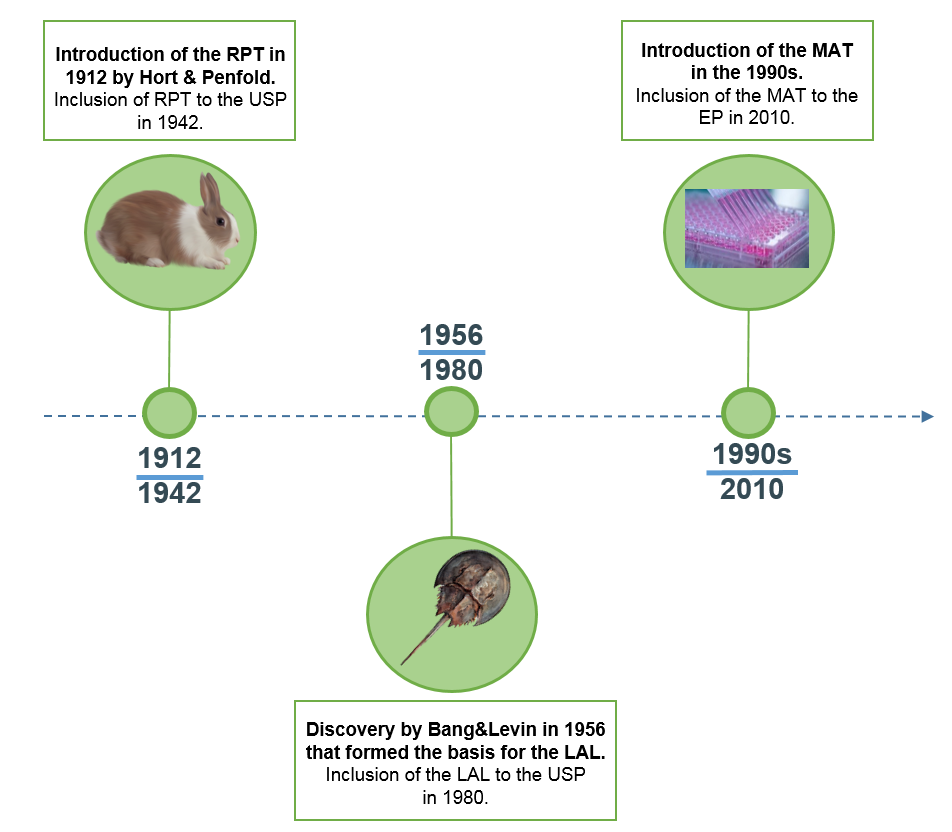
Test lal. Former India pacer and chief of the Cricket Advisory Committee (CAC) Madan Lal feels the presence of pace spearhead Jasprit Bumrah in the playing XI will give the visitors an edge going into the seriesdeciding fourth Test at the Gabba, Brisbane that gets underway on Friday. BioReliance offers the rabbit pyrogen test, in addition to the LAL test, as an alternative assay for the detection of endotoxin and other pyrogens The rabbit pyrogen test requires the injection of a small amount of batched test material into a rabbit’s blood stream, and monitoring for temperature increases. About LAL The in vitro Limulus Amebocyte Lysate (LAL) test was developed in the 1970s, as part of a 3Rs initiative to replace the need to use hundreds of thousands of rabbits each year in the testing of human and animal products for contamination LAL came to replace the Rabbit Pyrogen Test (RPT), which was established in the 1940s and was the goto safety test to measure fever reaction as an.
LAL Chromogenic endotoxin assay utilizes a modified Limulus Amoebocyte Lysate and a synthetic colorproducing substrate to detect endotoxin presence This assay is quantitative and the color intensity developed upon addition of the sample to the LAL supplied with the kit is proportional to the amount of endotoxin present in the sample and can be calculated from a standard curve. LALD is diagnosed by a blood test that measures the activity of the LAL enzyme Other supportive tests that your healthcare provider may order include genetic testing and a liver biopsy Biopsy results can help access liver damage, but cannot be used to diagnose LALD, since similar liver damage can occur from other diseases. The LAL test (acronym for Limulus Amebocyte Lysate) is a test for the determination of bacterial endotoxins, which uses an Amebocyte Lysate of the Limulus crab.
Endotoxin Testing (LAL Test) Endotoxin is a toxin that is released from gramnegative organisms, the tests determines whether these organisms are present (alive or dead) through the presence or lack thereof of those toxins. The LAL/TAL response to the test samples is compared to the response to the endotoxin standards in order to determine the amount of endotoxin in the test sample In the kinetic turbidimetric LAL/TAL method, a 96well plate reader or tube reader is used to monitor the change in solution clarity as a result of endotoxin present in a sample. The Bacterial Endotoxin test, also known as Limulus Amebocyte Lysate (LAL), is necessary to quantify this gramnegative bacteria within a cell wall Performed as a lot release test, the LAL assesses medical devices coming in contact with cerebrospinal fluid or the cardiovascular system.
The Bacterial Endotoxin test, also known as Limulus Amebocyte Lysate (LAL), is necessary to quantify this gramnegative bacteria within a cell wall Performed as a lot release test, the LAL assesses medical devices coming in contact with cerebrospinal fluid or the cardiovascular system. The LAL/TAL response to the test samples is compared to the response to the endotoxin standards in order to determine the amount of endotoxin in the test sample In the kinetic turbidimetric LAL/TAL method, a 96well plate reader or tube reader is used to monitor the change in solution clarity as a result of endotoxin present in a sample. Bio Products Laboratory Ltd;.
LALD is diagnosed by a blood test that measures the activity of the LAL enzyme Other supportive tests that your healthcare provider may order include genetic testing and a liver biopsy Biopsy results can help access liver damage, but cannot be used to diagnose LALD, since similar liver damage can occur from other diseases. Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on YouTube. LAL ECT funding must be used for inscope services and sites that will expand capacity for testing for COVID19 Funding may support a widerange of testing and testingrelated inscope activities that may change as COVID19 needs evolve within your community.
Limulus amebocyte lysate (LAL) test is commonly used for the detection of trace amounts of endotoxins (lipopolysaccharides, LPS) The mechanisms, LPS structural requirements and limitations of LAL tests are presented. Diagnose Wolman disease and cholesteryl ester storage disease (CESD) caused by LAL deficiency Limitations This test is not appropriate for determination of carrier status, as some clinical conditions may result in indeterminate enzyme activity levels. Our KineticQCL TM Kinetic Chromogenic LAL Assay provides a quantitative, kinetic chromogenic BET method and is the most sensitive of the LAL assays, with a sensitivity range of 0005 EU/ml 500 EU/ml The test sample is mixed with the chromogenic substrate in a 96well plate.
Activities to expand coronavirus testing capacity and access Activities must align with your LAL scope of project 2 Budget Narrative Clearly note if the proposed cost is a past cost (may be back to January ) 3 Equipment List Form (if applicable) 4 Other Relevant Documents (optional) Include your indirect cost rate agreement, if applicable. Genetic testing for family members and genetic prenatal diagnosis of pregnancies for women who are at increased risk are possible if family members carrying pathological mutations have been identified Management LAL deficiency can be treated with sebelipase alfa is a recombinant form of LAL that was approved in 15 in the US and EU. LAL test is a valuable test because it prevents the administration and or use of products and drugs which may cause fever, shock and death if highly endotoxic There are two methods of LAL endotoxin testing performed daily by Gibraltar’s highly experienced technicians GelClot method is based on the formation of clotting of the lysate in the.
Dr B Lal Clinical Laboratory has a wide range of 10 clinical laboratory tests You can choose tests like Complete Blood Counts test, ECG test, Liver Function Test, Kidney Function Test, Vitamin B12 test, Vitamin D25 test, Allergy test, and many more. Water Testing (AAMI/LAL) With a choice of LAL and AAMI water testing for your hemodialysis water, you can rely on AmeriWater’s fast, accurate and responsive service Along with an easytouse sampling kit, our customers receive free telephone consultation and can get results within 36 hours when necessary. Former India pacer and chief of the Cricket Advisory Committee Madan Lal feels the presence of pace spearhead Jasprit Bumrah in the playing XI will give the visitors an edge going into the seriesdeciding fourth Test at The Gabba India's injury woes have compounded with each passing day and the.
LAL Reagents Introduction to the LAL Test Limulus Amebocyte Lysate (LAL) tests detect and quantify bacterial endotoxins extracted from the outer membrane of gram negative bacteria The critical component of the LAL reagents used in endotoxin tests is derived from blood cells (amebocytes) of the horseshoe crab, Limulus polyphemus. ToxinSensor Single Test Kit is designed to be a qualitative In Vitro endpoint endotoxin test for human and animal parenteral drugs, biological products, and medical devices Limulus Amebocyte Lysate (LAL) as supplied is to be reconstituted with sample or control directly There is no need to reconstitute the system with LAL Reagent Water before use. Dr Lal path lab Test Packages Dr Lal Path Lab is also offering a wide range of health packages starting from Rs 799/ only Health packages have the advantage of cost and they test multiple parameters with the single sample We have a detailed post on Dr Lal Path Lab Packages which list all the tests and packages in details Avail Offer Now >>.
ChromoLAL Kinetic Chromogenic Formulation ChromoLAL lysate is lyophilized with substrate reagent and buffers It is optimized for the kinetic chromogenic LAL test method in microplate readers ChromoLAL is a buffered, stable and robust lysate, suitable for quantitative testing of a wide range of samples View Product. In a November 4, 1977, Federal Register notice (42 FR ), FDA described conditions for using LAL as a finished product test 6 By 19, FDA indicated in guidance that an LAL test could be. LAL ECT funding must be used for inscope services and sites that will expand capacity for testing for COVID19 Funding may support a widerange of testing and testingrelated inscope activities that may change as COVID19 needs evolve within your community.
Diagnose Wolman disease and cholesteryl ester storage disease (CESD) caused by LAL deficiency Limitations This test is not appropriate for determination of carrier status, as some clinical conditions may result in indeterminate enzyme activity levels. Test No Test Name 1 LAL Enzyme Assay (DBS or Whole Blood) Stat 2 LAL Gene Sequencing, performed only if Enzyme Assay is positivie (buccal or Whole Blood) Stat 3 Stat STAT is available for selfpay or institution accounts only, and reduces turnaround by >50% (less than 10 business days) for an additional 50% cost. L ysosomal A cid L ipase D eficiency LALD is a rare, genetic disease that causes progressive and ongoing damage Information for Healthcare Professionals Learn More Information for Patients And Caregivers.
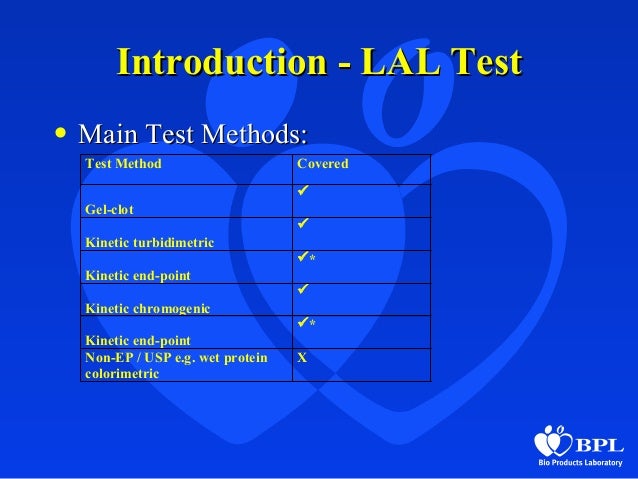
Gel Clot assay is a qualitative LAL test for detection of Gramnegative bacteria endotoxins The Gel Clot assay is run in tubes that are placed in a water bath or in dry heated oven at 37°C After a onehour incubation period, the tubes are flipped 180° A firm clot that stays in the bottom of the tube indicates a positive reaction. Dr Lal Pathlabs Packages in Southern, Western & Eastern India Dr Lal pathlabs has relatively new entrant in southern, western and eastern India Thus, to attract new customers & lower infrastructure cost, Dr Lal pathlabs packages are priced lower compared to northern India Here, the number of offering and test combinations are also more. LAL testing market is also segmented on the basis of testing methods into el clot endotoxin test, chromogenic endotoxin test & turbidimetric endotoxin test LAL Testing Market Country Level Analysis LAL testing market is analysed and market size insights and trends are provided by country, application & testing methods as referenced above.
Dr Lal PathLabs Quality and Service is now available at your fingertips via our Mobile App which has been made with utmost care and the objective of keeping it simple yet effective Key features of the app include Book a Home Collection Now you can book a Blood test or a Urine test right from the comfort of your Home with a few clicks Before booking a diagnostic lab test, you can also. Water Testing (AAMI/LAL) With a choice of LAL and AAMI water testing for your hemodialysis water, you can rely on AmeriWater’s fast, accurate and responsive service Along with an easytouse sampling kit, our customers receive free telephone consultation and can get results within 36 hours when necessary. L ysosomal A cid L ipase D eficiency LALD is a rare, genetic disease that causes progressive and ongoing damage Information for Healthcare Professionals Learn More Information for Patients And Caregivers.
The Thermo Scientific Pierce LAL Chromogenic Endotoxin Quantitation Kit measures the amount of endotoxin in a protein, peptide or antibody sample using the Limulus Amebocyte Lysate (LAL) assayFeatures of the LAL Chromogenic Endotoxin Quantitation Kit Sensitivedetect as little as 01 EU/mL (approx. Several variations of the LAL assay exist for different applications The simplest form is the LAL gel clot assay, which monitors a piece of LAL gel for clotting upon exposure to a test sample More advanced techniques rely on turbidimetric or chromogenic methods for quantification. Limulous amoebocyte lysate (LAL) is the test performed as this is based in the biology of the horseshoe crab which produces LAL enzymes in blood cells to bind and inactivate endotoxin from invading bacteria This was accepted by the US FDA as an endotoxin test method in 19.
51 Precaution to be taken during LAL testing 511 Check the calibration status of the triobloc 512 Check the temperature of triobloc before keeping the sample tubes into the wells, it should be 37±1°C 513 All ready to use glassware / accessories must have proper certification for endotoxinfree quality. Bacterial Endotoxin (LAL) Testing Bacterial LAL Testing STERIS provides contract analysis of bacterial endotoxins using methods compliant with EP, USP and ANSI/AAMI ST72 to meet FDA and MHRA requirements Testing is performed according to USP , USP and ANSI/AAMI ST72. Limulus Amoebocyte Lysate Test The limulus amoebocyte lysate (LAL) test is a simple method for the detection of viable and nonviable Gramnegative bacteria Certain cellwall lipopolysaccharides (ie endotoxins) of this bacterial group lead to gelation of blood cell (amoebocytes) lysates of the Limulus polyphemus crab Using a dilution row.
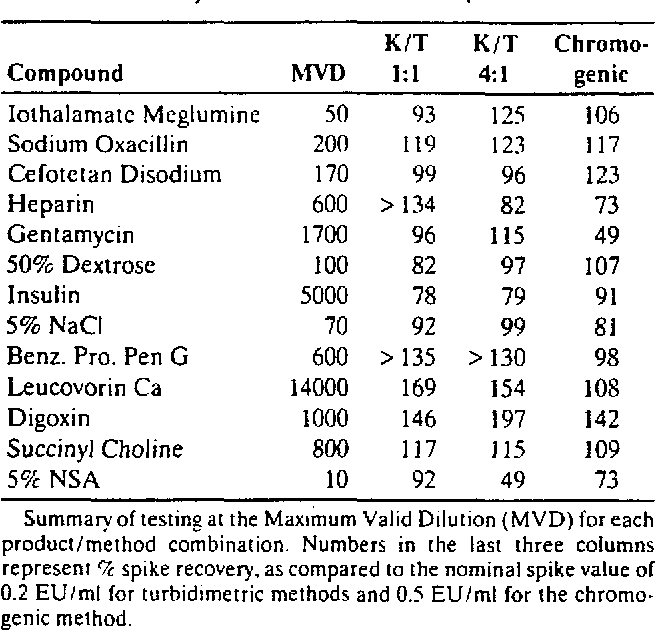
The LAL (limulus amebocyte lysate) testing, also known as bacterial endotoxin testing, is an in vitro assay used to detect the presence and concentration of bacterial endotoxins in drugs and biological products, and is an important part of pharmaceutical microbiology. According to a Parenteral Drug Association (PDA) survey "Current Practices in Endotoxin and Pyrogen Testing in Biotechnology" (55), as of 1990 the gelclot assay was the most used assay (77%) with the remainder, the kinetic chromogenic and turbidimetric LAL assays, representing less than onethird of users responding Most of respondents (71%) indicated for the continued use of the rabbit. Variability in the LAL test has been studied and many have been documented Sources of variation include formulations of LAL reagent and control standard endotoxin, container effects on endotoxin preparation, laboratory materials and equipment, lottolot product variation, and LAL test method ( 1317) The following.
ChromoLAL Kinetic Chromogenic Formulation ChromoLAL lysate is lyophilized with substrate reagent and buffers It is optimized for the kinetic chromogenic LAL test method in microplate readers ChromoLAL is a buffered, stable and robust lysate, suitable for quantitative testing of a wide range of samples View Product. This test is used to detect endotoxin in a given substance using amebocyte lysate from horseshoe crab 3 different methods used to detect endotoxins are also ex. Limulus Lysate Test LAL tests are only valid in situations where standard endotoxin can be shown to be detectable with the same efficiency in a test sample as in a control consisting of water (LAL reagent water, LRW) known to be endotoxin free (LRW) From Pharmaceutical Microbiology, 16.
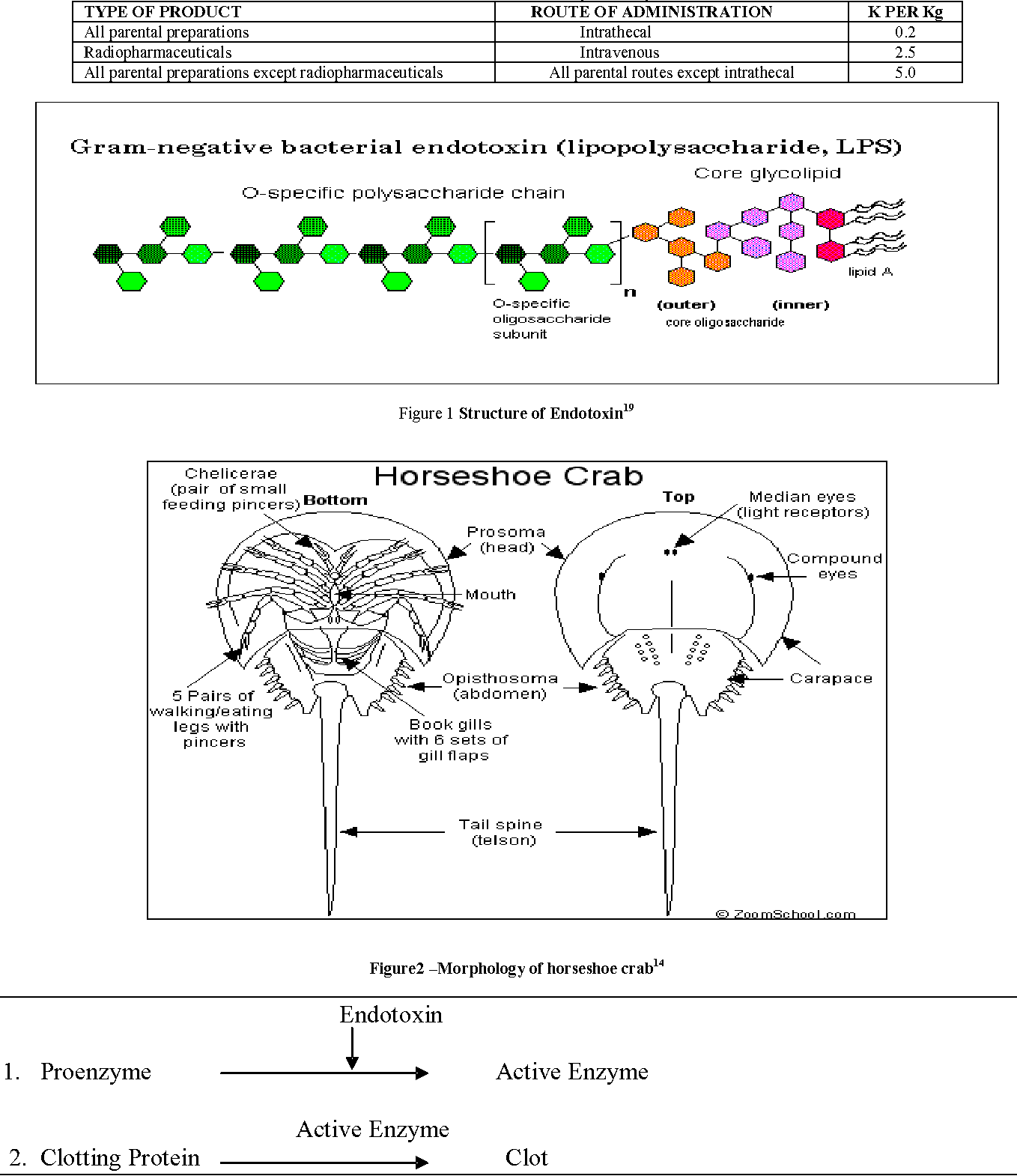
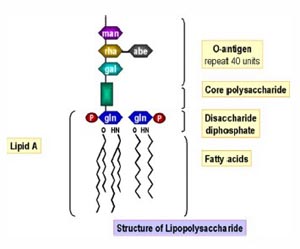
Variability in the LAL test has been studied and many have been documented Sources of variation include formulations of LAL reagent and control standard endotoxin, container effects on endotoxin preparation, laboratory materials and equipment, lottolot product variation, and LAL test method ( 1317) The following. Atlantic horseshoe crab ' Limulus polyphemus blood used for the Test Limulus amebocyte lysate (LAL) is an aqueous extract of blood cells (amoebocytes) from the Atlantic horseshoe crab Limulus polyphemus LAL reacts with bacterial endotoxin lipopolysaccharide (LPS), which is a membrane component of gramnegative bacteria.

Lal And Tal In The Us Pharmacopoeia

Endotoxin Lal Test Kit Bacteria Detection Kit ज व ण पर क षण क ट yur Lifesciences Mumbai Id
The Principle Of Limulus Amebocyte Lysate Lal Based Electrochemical Download Scientific Diagram
Horseshoe Crab Research Kepley Biosystems Inc

Results Of Pharmaceutical Products By The Lal Gel Clot And Chromogenic Download Table
An Overview Of Limulus Amoebocyte Lysate Lal Test Semantic Scholar

Endotoxin Lal Test Kit Supplier Distributor In Kalyan Maharashtra

The Detection Of Endotoxins Via The Lal Test The Chromogenic Method

Endotoxin Testing

Lal Test And Rpt For Endotoxin Detection Of Cpt 11 Dspe Mpeg00 Nanoformulation What If Traditional Methods Are Not Applicable Sciencedirect
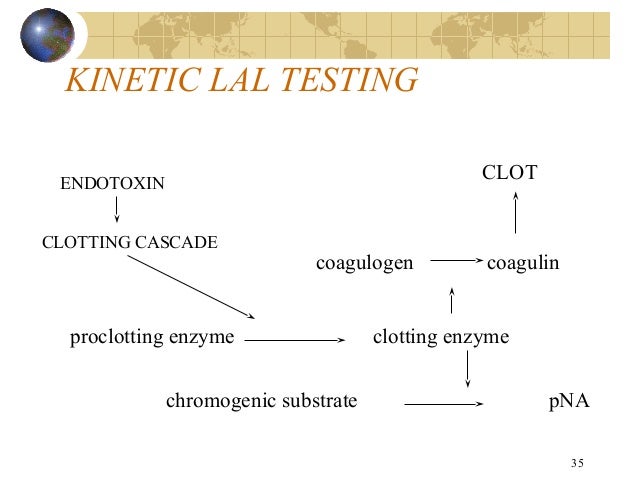
Lal Presentation
Q Tbn And9gcqv61 8bayhtv3fjaoc3 2opj555yrd357prcj6104wvkphljb0 Usqp Cau

Blood Test For All Type Dr Lal Path Labs In Amritsar

Pdf Challenges On Performing Lal Test In Oil Products Milan B Arambasic Academia Edu

Full Text Utility Of An Automatic Limulus Amebocyte Lysate Kinetic Turbidimetric Mder
Pdf Variability In The Lal Test Comparison Of Three Kinetic Methods For The Testing Of Pharmaceutical Products Semantic Scholar
Mat Regulations

Bacterial Endotoxin Testing Bet Lal Test Helvic Laboratories
Lal Limulus Amebocyte Lysate The Horseshoe Crab
Covid 19 Antibody Detection Test Dr Lal Pathlabs

Lal Limulus Amebocyte Lysate The Horseshoe Crab

Evaluating The Costs Of Endotoxin Testing Promega Connections

History Of The Lal Test Validation And Regulatory Acceptance Article In German Altex Alternatives To Animal Experimentation
Government Has Asked Private Hospitals To Ready 000 Beds For Coronavirus Patient Dr Arvind Lal Lal Pathlabs Zee Business

Pdf Variability And The Lal Assay

Pyrogen Testing As Per Ip Bp Usp

Ppt Limulus Amebocyte Lysate Lal Test Methods Powerpoint Presentation Id
Bacterial Endotoxin Test

Schematic Study Sequence Abbreviations Lal Water Water With An Download Scientific Diagram

Gel Clot Technique Turbidimetric Technique Chromogenic Technique Wako Lal System

Dr Lal Pathlabs Book Immunity Check Package To Test How Prepared Is Your Body To Fight Against Infections Beahero And Keep You And Your Family Safe From The Chance Of
Lipopolysaccharides Lps Elisa Kit May Substitute For Limulus Amebocyte Lysate Lal Test Reagent Cloud Clone Corp Ccc

Lal Testing Authorstream

Limulus Amebocyte Lysate Lal Test Methods Ppt Download

Endotoxin Testing And The Horseshoe Crab What S Next

Coronavirus Symptoms Testing Centre Near Me Faq Coronavirus Dr Lal Pathlabs
Pharmaceutical Microbiology Gel Clot Test

Dr Lal Pathlabs After Dr Lal Pathlabs Here S The Rx On Healthcare Data Security Scan Test And Treat Immediately The Economic Times

Lal Test Important For Exam Limulus Amebocyte Lysate Test Of Parenteral Youtube

New Publication About Bacterial Endotoxin Testing Available Microcoat De

Gel Clot Lyophilized Amebocyte Lysate Multi Test Vial Manufacturers And Suppliers Price Bioendo Technology

Pdf In This Issue Choosing An Lal Test Method Cse Activity Master Card Visa Calendar A Wealth Of Options Choosing An Lal Test Method Stanislaw Yohan Academia Edu

Your Covid 19 Test Result Likely Exposed Dr Lal Pathlabs Risked Patient Data Ibtimes India

Bio Resource Bacterial Endotoxin Test Lal Test Principle Advantages

Comparing The Lal Method With The Pyrogen Test On Rabbits

Pyrogen Testing
Chromogenic Lal Assay Lonza
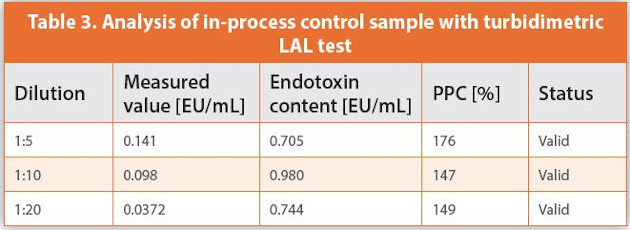
The Truth Of Endotoxin Values Points For Consideration During Investigation Of Aberrant Bet Results American Pharmaceutical Review The Review Of American Pharmaceutical Business Technology

Overview Of Lal Test Technology Endotoxin Testing Lipopolysaccharide Life Sciences

Comparative Evaluation Of Pyrogens Tests In Pharmaceutical Products
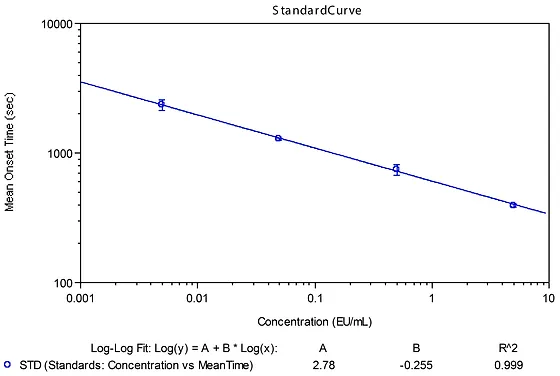
Lal Chromogenic Endotoxin Test Creativescientist

Lal Test To Confirm The Contamination Of Endotoxin Download Table

Dr Lal Path Labs Price List Flat 3 Cashback Book Online Labsadvisor

Test Code Dr Lal Pathlabs Blog
2

Recombinant Factor C Assay To Aid Demand For Lal Endotoxin Testing

Private Labs Corona Test Dr Lal Pathlabs Is Ready To Conduct Coronavirus Test If Govt Allows Cfo Ved Goel Cfo News Etcfo

Cell Culture Faqs Bacterial Endotoxin Contamination Sigma Aldrich

Lal Test For Endotoxin Youtube

Limulus Amebocyte Lysate Lal Test Methods Ppt Video Online Download
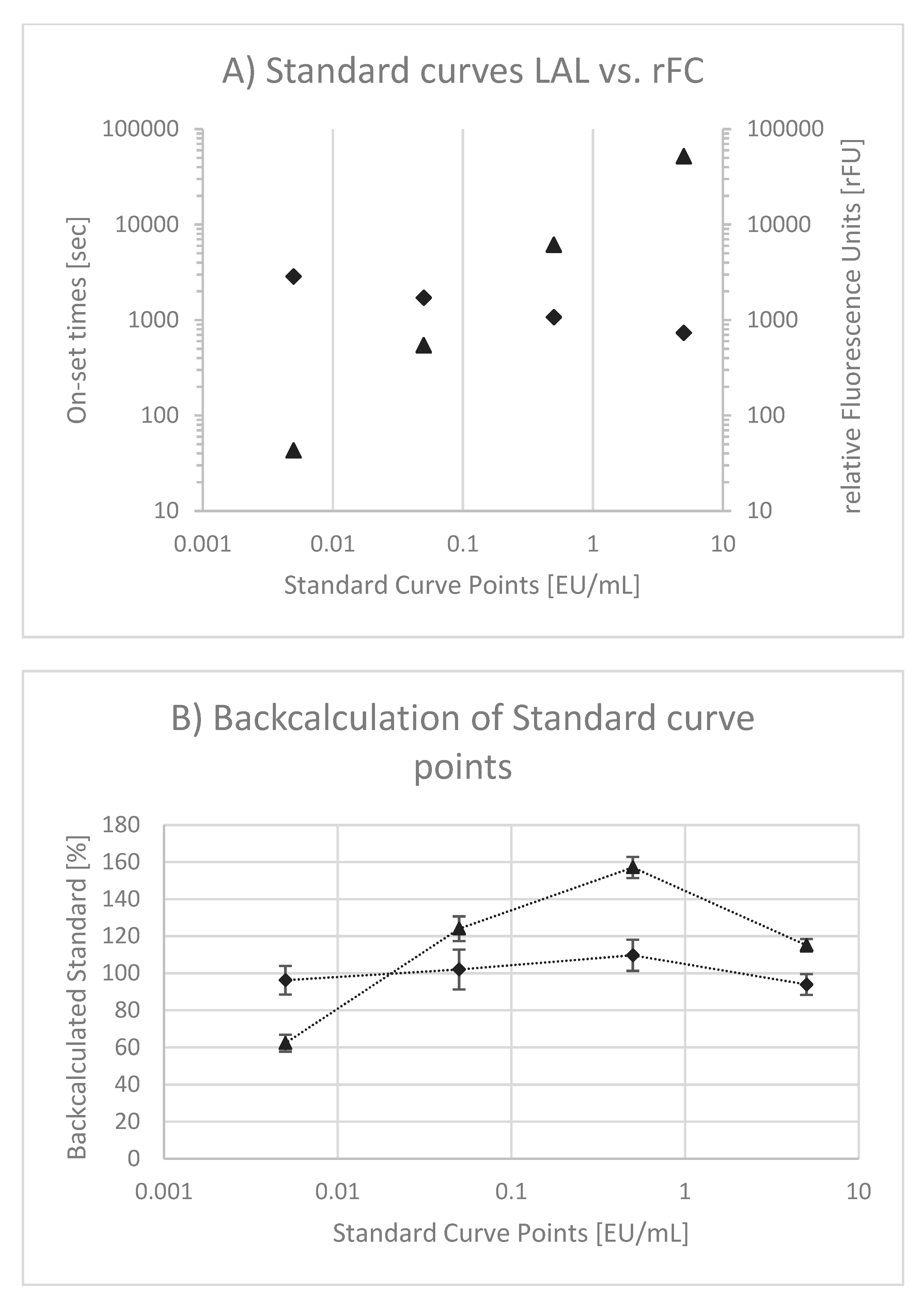
Microorganisms Free Full Text Comparison Of Lal And Rfc Assays Participation In A Proficiency Test Program Between 14 And 19 Html
Dr Lal Pathlabs Apps On Google Play

Limulus Amoebocyte Lysate Lal Test Download Scientific Diagram

Thyrocare Tech Dr Lal Path Labs Surge As Pvt Labs To Conduct Covid 19 Test Business Standard News

Lal Limulus Amebocyte Lysate Endotoxin Test By Acronymsandslang Com

Limulus Amoebocyte Lysate Lal Test For Pyrogen Detection Microbiology Class
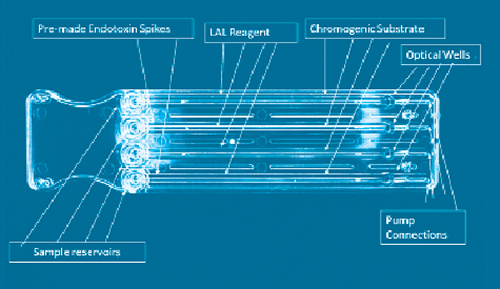
Screening Successfully For Endotoxins
8 5 Test For Bacterial Endotoxins ตำรายาของประเทศไทย กรมว ทยาศาสตร การแพทย

Dr Lal Pathlabs Again A Multibagger In Future Pa Wealth Advisors Blog
Q Tbn And9gcsjv8cfin64jxdlbyhaqzeumpmg3xdzrd3n9ojzzya5fx5pbndp Usqp Cau

The Detection Of Endotoxins Via The Lal Test The Gel Clot Method Pyrostar

Bacterial Endotoxin Lal Test Kit At Rs Kit Bacteria Test Kits Id

Bacterial Endotoxin Testing Lal Charles River

Glucose Challenge Test In New Delhi From Dr Lal Pathlabs Ltd View Price Interpretations Reviews 1mg

Dr Lal Pathlabs Leaks Millions Of Patients Data In Public Domain Report Technology News The Indian Express
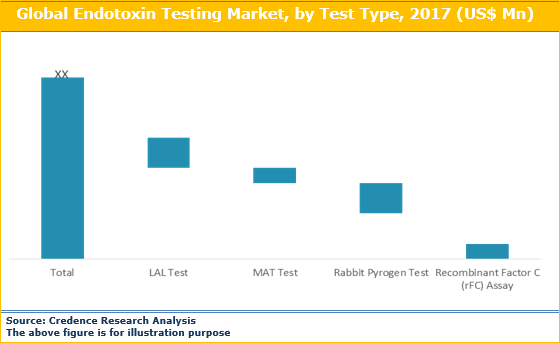
Endotoxin Testing Market Size Share Trend And Forecast To 26

Cortisol In New Delhi From Dr Lal Pathlabs Ltd View Price Interpretations Reviews 1mg

Bacterial Endotoxin Test Wako Lal System

Endotoxin Or Lal Test Eurofins Deutschland
Http Bioscreen Com Images Technicalbulletins Tb01 Endotoxin Pdf
2
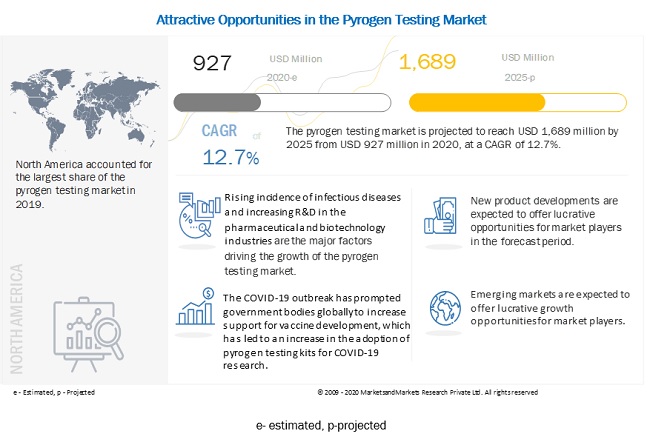
Pyrogen Testing Market Global Forecast To 25 Marketsandmarkets
Gel Clot Lal Assay Lonza
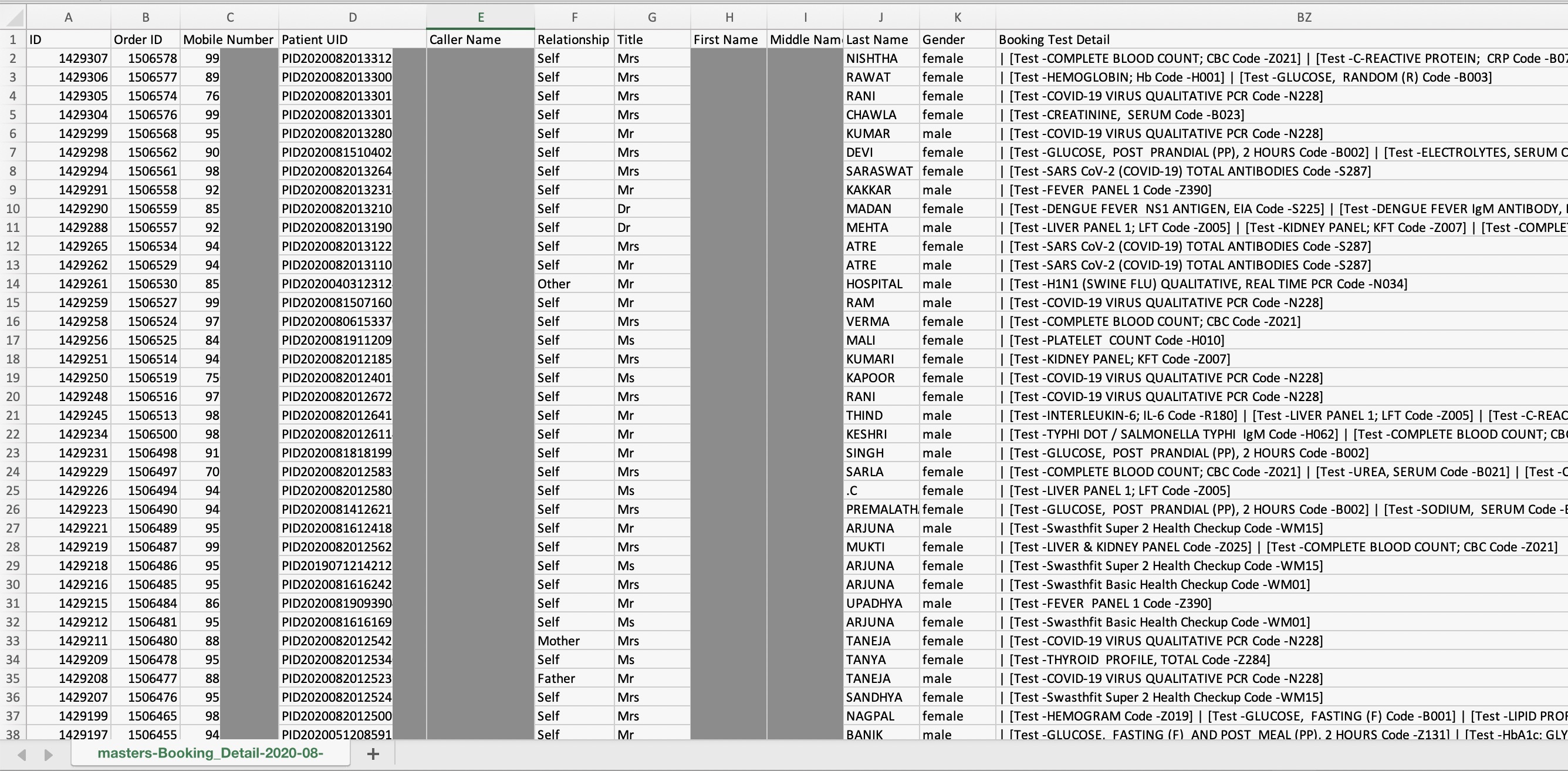
Dr Lal Pathlabs One Of India S Largest Blood Test Labs Exposed Patient Data Techcrunch

Full Text Utility Of An Automatic Limulus Amebocyte Lysate Kinetic Turbidimetric Mder

The Necessity Of Lal Endotoxin Testing Pyrsotar Medical Laboratory Laboratory Biology Art
Q Tbn And9gctcupymrwaa7lrxjluerlmrigbx6axbyvzuqcrfxqz Czfqhckk Usqp Cau
Dr Lal Pathlabs Chembur East Pathology Labs In Mumbai Justdial

Result Of The Test For Lal Download Table

Top Pdf Limulus Amebocyte Lysate Lal Test 1library
Resolved Dr Lal Pathlabs Delay In Reports And Incomplete Information From Customer Care Executive

Buy Good Price Limulus Amebocyte Lysate Assay Lal Reagent Single Test Vial Gel Clot Method From Xiamen Bioendo Technology Co Ltd

Accessories Suitable For Carrying Out The Lal Test Pyrostar

What Is Limulus Amebocyte Lysate Lal And Its Applicability In Endotoxin Quantification Of Pharma Products Intechopen
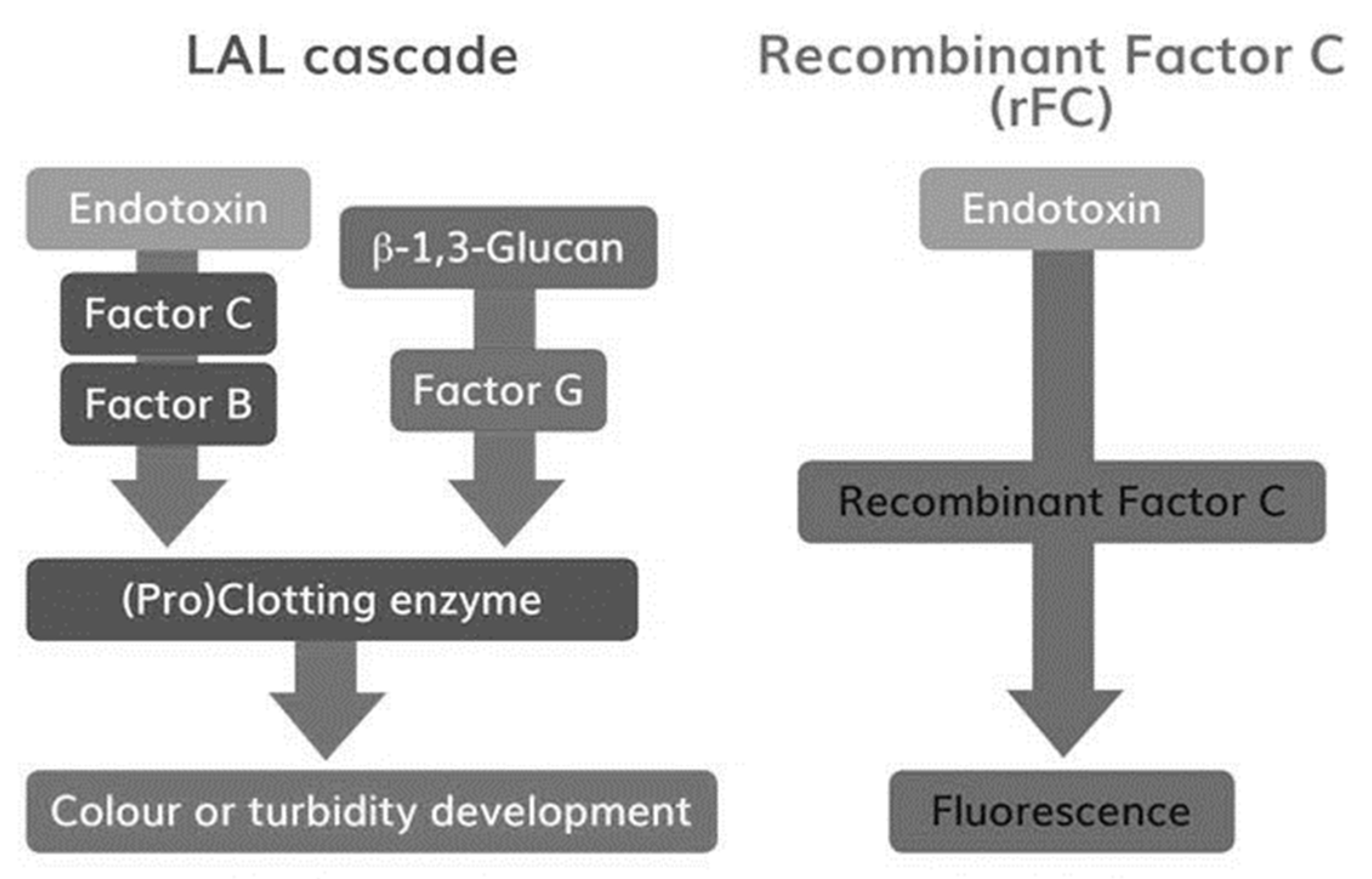
Microorganisms Free Full Text Comparison Of Lal And Rfc Assays Participation In A Proficiency Test Program Between 14 And 19

Bacterial Endotoxin Test

Bacterial Endotoxin Test
Q Tbn And9gctb36tctwfcdn3mjyi5aedjeqjyh4kkcfap8a7 J9akbdanwfkn Usqp Cau



